
Companies continue to share news about the data that will be presented at the 2024 ASRS meeting in Stockholm, Sweden.

Companies continue to share news about the data that will be presented at the 2024 ASRS meeting in Stockholm, Sweden.

A novel formulation may prevent blood vessel growth and vascular leakage in the retina.

Ophthalmologist receives the Roger Steinert, MD, Legacy Award during the Octane Ophthalmology Tech Forum in Irvine, California.
A team of researchers at the Ohio State University found that in mice an enzyme related to cell growth and division is a culprit in the blood vessel invasion in the back of the eye that causes blurred central vision in wet AMD.
The company announced it has received FDA approval to relaunch Susvimo for intravitreal use via an ocular implant for treating people with wet AMD.
Prescriptions with semaglutide (Wegovy, Ozempic, Novo Nordisk) are FDA approved to treat obesity and type 2 diabetes, respectively.

The funding will go toward the continued clinical development of the company’s lead asset, AGTC-501 (laruparetigene zovaparvovec)

The pharmaceutical candidate is being evaluated as a potential treat for nAMD.

42nd Annual Meeting in Stockholm, the Venice of the North
According to Genentech, faricimab-svoa is the first and only syringe prefilled with an FDA-approved bispecific antibody for the treatment of wet AMD, DME and RVO.

The deal included a total upfront consideration of $81 million, including a cash payment of approximately $65 million.

Apellis plans to seek re-examination, with a final opinion expected in fourth quarter of 2024.
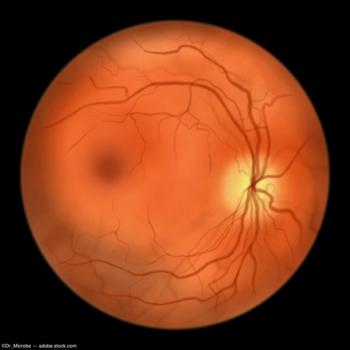
Faricimab is already approved in the EU and UK for neovascular age-related macular degeneration and diabetic macular edema.

The company expects approval from the EMA by 2025.
Repeated 0.1-ml intravireal injections of lampalizumab, antigen-binding fragment of a humanized monoclonal antibody that inhibits complement factor D, seem to be associated with a low risk of elevated IOP
The biosimilar was approved for treating patients with age-related neovascular (wet) macular degeneration (nAMD) and other serious retinal diseases.

During interview earlier this year, we asked leaders in the retina field would tell residents and medical students as they train for the next decades in retinal care. Here's what 2 of them had to say.

We asked what leaders in the retina field would tell residents and medical students as they train for the next decades in retinal care. Here's what 2 of them had to say.

From AI tools and early diagnoses to advancing image rendering and predicting overall health, retinal imaging is reaching new heights.

The ASRS annual meeting will take place in Stockholm, Sweden on July 16-20, 2024. Several major updates are already anticipated at the event.

4D-175 will be evaluated in the GAZE clinical trial, an upcoming phase 1, open-label trial.

The award, supported by Oculis, will recognize outstanding contributions to retina research and will grant €30,000 for research support, and an additional €5,000 directly to the candidate.

The diagnostic and monitoring tools use artificial intelligence-based models.

VUMC noted that the diagnosis of diabetes and treatment needed to prevent diabetes complications may be delayed in people who carry the variant, G6PDdef.

Peter McDonnell, MD, shares his thoughts on Omar R. Shakir's, MD, MBA, article on office-based vitrectomy surgery.

According to the company, the acquisition includes 2 commercial assets Iluvien and Yutiq, expanding ANI’s foothold in ophthalmology.

The 2024 event was held in Seattle, Washington, from May 5 through May 9. Members of the Ophthalmology Times team attended and interviewed several researchers and industry professionals, highlighting news from the meeting.
The results were presented at a symposium during the American Diabetes Association's 84th Scientific Sessions in Orlando, Florida, and mark the first large-scale trial specifically designed to investigate the effect of fenofibrate on eye outcomes in people with early diabetic retinopathy.

OCU410ST is a modifier gene therapy candidate being developed for Stargardt disease, which affects approximately 100,000 people in the United States and Europe combined.